
Web site: HOMO (Molecular Orbital) - an overview | ScienceDirect Topics.DFT - gaussviev - gaussian09W - HOMO - LUMO. These are also commonly referred to as HOMO-1 and LUMO+1, respectively. GAUSSIAN 09W TUTORIAL AN INTRODUCTION TO COMPUTATIONAL CHEMISTRY USING G09W. They are named NHOMO for next-to-highest occupied molecular orbital and SLUMO for second lowest unoccupied molecular orbital. If existent, the molecular orbitals at one energy level below the HOMO and one energy level above the LUMO are also found to play a role in frontier molecular orbital theory. HOMO and LUMO are acronyms for highest occupied molecular orbital and lowest unoccupied molecular orbital, respectively. This abbreviation may also be extended to semi occupied molecular orbital. The HOMO of each CH/N substituted molecules is mainly localized on residual phenoxide side of the ligand A with a relatively significant contribution from the nitrogen atom. The HOMO is the highest energy MO that has any. The HOMOs and LUMOs in the CH/N substituted derivatives of mer -Alq3 show the same trend of localization at A and B ligands, respectively. the shift in energies and that in the D3h structure the HOMO and LUMO. These are: the highest occupied molecule orbital or HOMO and the lowest un-occupied molecular orbital or LUMO. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators. SOMOĪ SOMO is a singly occupied molecular orbital such as half-filled HOMO of a radical. the MO diagram for linear water HOH, using this diagram explain if linear. In organometallic chemistry, the size of the LUMO lobe can help predict where addition to pi ligands will occur. The same analogy can be made between the LUMO level and the conduction band minimum. The HOMO level is to organic semiconductors roughly what the maximum valence band is to inorganic semiconductors and quantum dots. The difference in energy between these two frontier orbitals can be used to predict the strength and stability of transition metal complexes, as well as the colors they produce in solution. HOMO and LUMO are sometimes collectively called the frontier orbitals, such as in the frontier molecular orbital theory. The energy difference between the HOMO and LUMO is termed the HOMO–LUMO gap. 
The acronyms stand for highest occupied molecular orbital and lowest unoccupied molecular orbital, respectively. It has been explained to me I just need to measure the mV difference between the peak of the ferrocene and the oxidation or reduction curve, then subtract/add that onto the reference for ferrocene (4.8eV) to get the HOMO/LUMO levels.In chemistry, HOMO and LUMO are types of molecular orbitals. If we extend this reasoning to two interacting molecules we realize that the outer filler orbital ( the Highest Occupied Molecular Orbital, HOMO) will react. My CV measurements result in a current-voltage curve (IV curve). I am interested in determining the HOMO and LUMO levels of the material. I've carried out CV measurements on a few different types of material (with a ferrocene reference). Therefore, the FERMO concept is an alternative way to explain chemical phenomena when HOMO-LUMO arguments fail or cannot be applied. explaining why DFT+ models could predict the electronic conductance in these. I'm a physicist, so I apologize if these are obvious questions. functional typically underestimate molecular HOMO-LUMO gaps by several eVs. Can the sweep rate change the observed redox potential? Yes.Could the oxidized or reduced species undergo reactions that change the observed redox potential? Yes.Could the molecules aggregate and change the observed redox potential? Yes.Can the electrode change the observed redox potential? Yes.Can the solvent change the observed redox potential? Yes.There should be a correlation between oxidation and reduction potentials and HOMO and LUMO energies.īut step back and think - in one case, you have electrochemistry in the electrolyte solution at an electrode surface. 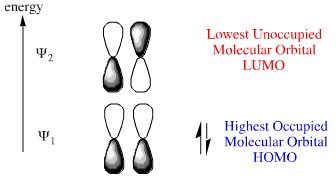
When you sweep the potential with CV, the cathodic peak ($E_$$Īre these peaks even the HOMO/LUMO levels at all? A conjugated system has a region of overlapping p-orbitals, bridging the interjacent single bonds, that allow a delocalization of electrons across all the. Note that the "peak" actually has two sides. As in the previous examples the pi system moves from a LUMO species to a HOMO species, meaning this reaction is allowed. If the ring opening uses a conrotatory process then the reaction results with the HOMO of butadiene. Let's start with interpreting the cyclic voltammetry curves themselves. a constructive bond) and the LUMO of the pi bond are important in the FMO theory consideration. Yes, you can "convert" this way, but you're correct to be skeptical. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
